Shijiazhuang Mingxu Chemicals Co., Ltd. is a professional specialty chemicals company in China and started in 2008.It emerged as a professional pharmaceutical company by supplying high standard APIs, pharmaceutical intermediates especially used for cephalosporins, finished formulations meeting BP, USP, JP, EP and customer specifications.
Why choose us?
Experience
With more than 10 years of industry experience, we have an in-depth understanding of the field of polyurethane catalysts. Our expertise allows us to develop innovative solutions that meet our customers' specific requirements. We have successfully served various industries including construction, furniture, shoe sole, automotive, coatings, etc.
Product
Our comprehensive product range addresses different applications and customer needs. We offer a variety of catalysts that enhance the performance and characteristics of polyurethane products. These include amine-based catalysts, metal-based catalysts and specialty catalysts customized for specific applications. Our products are continually reviewed and improved to ensure optimal results and compliance with industry standards.
Team
Our talented and dedicated team is instrumental in driving our company's success. We have a team of experienced chemists and engineers who are passionate about their work. Their expertise coupled with their commitment to continuous learning and innovation enables us to provide our customers with cutting-edge products and tailor-made solutions.
Quality
We have established a strict quality management system to manage every aspect of our operations, from raw material procurement to product manufacturing and delivery. We adhere to the highest quality standards and use advanced testing methods to ensure our catalysts meet all relevant specifications, including purity, reactivity and stability. Our commitment to quality doesn't end with our products, as we also prioritize excellent customer service and timely delivery.
-
MXC-EHATA:64485-82-1BRAND NAME:EHATA. PRODUCT NAME: ETHYL(Z)-2(2-AMINOTHIAZOLE-4-YL) HYDROXYIMINO ACETATE. CAS NO.: 64485-82-1. FORMULA:C7H9N3O3S. Assay: ≥98%. Purity: ≥98.5%read more
-
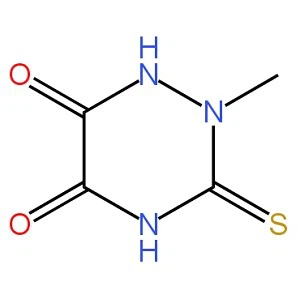
MXC-THIOTRIAZINONEBRAND NAME:TTZ. PRODUCT NAME:Mercapto-2-Methyl-5-oxo-6- Hydroxy-1,2,4-Triazine. CAS NO.:58909-39-0. FORMULA:C4H5N3O2S . Assay:Min.98.5% . Purity: Min. 99.0%read more
-
MXC-CEFTAZIDIME INTERMEDIATEBRAND NAME:TAEM. PRODUCT NAME:S-2-Benzothiazolyl (Z)-2-(2-aminothiazol-4-yl)-2- (1-t-butoxycarbonyl-1-methyl)ethoxyiminothioacetate. CAS NO.:158183-05-2. FORMULA:C20H22N4O4S3. Purity: ≥ 98.5%. Assay:98.0%read more
Pharma intermediate refers to a chemical compound that is produced during the manufacturing process of a pharmaceutical product. It is a key intermediate product that is used in the production of a drug or active pharmaceutical ingredient (API). These intermediates are synthesized from starting materials and then further processed to produce the final drug product. They are important in the pharmaceutical industry because they can significantly influence the quality, efficacy, and safety of the final drug product.
Pharmaceutical intermediates are compounds that are used as building blocks in the synthesis of active pharmaceutical ingredients (APIs). They are essential components in the production of drugs and play a critical role in the pharmaceutical industry. In this article, we will discuss the importance of pharmaceutical intermediates, their types, and their uses.
Importance of Pharmaceutical Intermediates:
Pharmaceutical intermediates are crucial in the drug development process. They are used to synthesize APIs that are used to produce finished pharmaceutical products (FPPs). The quality and purity of intermediates are essential in ensuring the efficacy and safety of the final product. Therefore, the development and production of intermediates must adhere to strict regulations and quality standards.

Types of Pharmaceutical Intermediates
Pharmaceutical intermediates can be classified into various types based on their chemical structure, function, and source. Some of the common types of intermediates include:
Amino Acids And Peptides
Amino acids and peptides are important intermediates in the synthesis of protein-based drugs. They are used in the production of antibiotics, hormones, and vaccines.
Nucleotides
Nucleotides are used in the synthesis of nucleic acid-based drugs, such as DNA and RNA-based therapeutics.
Carbohydrates
Carbohydrates are used in the production of vaccines and other biologics. They are also used as excipients in FPPs.
Steroids
Steroids are intermediates used in the synthesis of hormone-based drugs.
Heterocyclic Compounds
Heterocyclic compounds are intermediates used in the production of a wide range of drugs, including antidepressants, antivirals, and anticancer agents.
Uses of Pharmaceutical Intermediates
Pharmaceutical intermediates are used in various stages of drug development, including discovery, preclinical testing, and clinical trials. They are also used in the production of FPPs. Some of the common uses of pharmaceutical intermediates include:
Drug Discovery: Pharmaceutical intermediates are used to synthesize new chemical entities (NCEs) that can be screened for potential drug candidates.
Preclinical Testing: Intermediates are used to test the efficacy and toxicity of potential drug candidates in animal models.
Clinical Trials: Intermediates are used to produce APIs for use in clinical trials.
Production of FPPs: Intermediates are used to produce APIs that are used in the production of FPPs.
Pharmaceutical intermediates play a crucial role in the development and production of drugs. They are essential components in the synthesis of APIs and are used in various stages of drug development. The quality and purity of intermediates are important in ensuring the efficacy and safety of the final product. Therefore, the development and production of intermediates must adhere to strict regulations and quality standards.
The Difference Between Pharmaceutical Intermediates And Apis
Pharmaceutical intermediates / APIs
Pharmaceutical intermediates and APIs both belong to the category of fine chemicals.
Pharmaceutical intermediates are some chemical raw materials or chemical products used in the synthesis process of APIs.APIs are products that have completed the synthesis pathway and are used as an active ingredient in pharmaceuticals.
Intermediates can only be used to synthesise the next step in the product pathway, through which the API is manufactured; whereas APIs can be directly formulated.
The production of APIs requires a manufacturing licence. Pharmaceutical intermediates, on the other hand, do not require a production licence and can be produced in ordinary chemical plants and used in the synthesis of APIs as long as they reach a certain level.Because of this, the threshold of entry for pharmaceutical intermediates is relatively low and the competition is relatively fierce.
Then, to buy pharmaceutical intermediates among many suppliers, besides looking at the price, we also have to look at the quality, scale and management level of the enterprise.
What are APIs?


API intermediates are the core components of pharmaceutical products that provide therapeutic effects. An API intermediates is a chemically active substance that is responsible for the intended pharmacological activity of a medication. These ingredients are typically synthesized through chemical processes or derived from natural sources.
The role of APIs intermediates in pharmaceutical products is crucial. They are responsible for the therapeutic effect of the medication, meaning they directly interact with the target in the body to produce a desired physiological response. APIs are carefully selected based on their pharmacological properties and their ability to treat a specific medical condition.
APIs are crucial for effective, safe, and high-quality medications. They undergo rigorous testing and regulatory approval to ensure safety and efficacy. Proper identification, purity, and potency are vital for medication quality.
There are numerous examples of commonly used APIs in medications. Here are a few examples:
Paracetamol (Acetaminophen): It is a widely used analgesic (pain reliever) and antipyretic (fever reducer).
Ibuprofen: This nonsteroidal anti-inflammatory drug (NSAID) is used to alleviate pain, reduce inflammation, and lower fever.
Omeprazole: It is a proton pump inhibitor (PPI) used to treat conditions like gastroesophageal reflux disease (GERD) and stomach ulcers.
Simvastatin: This API belongs to the statin class of drugs and is used to lower cholesterol levels in the blood.
Ceftazidime intermediate is a chemical compound that is produced during the synthesis of the antibiotic ceftazidime. It is a critical component in the manufacturing process of ceftazidime, which is used to treat bacterial infections. Ceftazidime intermediate is typically a complex mixture of several compounds, and its composition can vary depending on the specific manufacturing process used. It is not used directly in medical treatments and is only used in the production of ceftazidime.
Application of Ceftazidime Intermediate
The intermediate of ceftazidime, (6R, 7R)-7-[[(Z)-2-(2-Amino-4-thiazolyl) acetyl] amino]-3-[[(1-carboxy-1-methylethoxy) imino] methyl]-8-oxo-5-thia-1-azabicyclo [4.2.0] oct-2-ene-2-carboxylic acid, has various applications in pharmaceutical and chemical industries. Some of the significant uses of ceftazidime intermediate are:

Synthesis of Antibiotics
Production of Antibacterial Drugs
Biological Applications
Chemical Industry
Research Applications
Ceftazidime intermediate has an important role in the production of antibiotics and is widely used in the pharmaceutical and chemical industries.
Synthetic Methods of Ceftazidime Intermediate And Ceftazidime
There are various synthetic methods for the production of ceftazidime intermediate and ceftazidime.
Synthesis of Ceftazidime Intermediate:
Nitration: The first step is the nitration of 7-aminocephalosporanic acid (7-ACA) to form 7-nitrocephalosporanic acid (7-NC). This is carried out by reaction with nitric and sulfuric acid.
Reduction: The 7-nitrocephalosporanic acid is then reduced to 7-aminocephalosporanic acid using hydrogen and a catalyst such as palladium on carbon.
Acylation: The 7-aminocephalosporanic acid is then acylated with 4-methyl-2-pyridinemethanol and benzyl bromide to form the ceftazidime intermediate, N-[(6R,7R)-7-[[(2Z)-2-(2-amino-4-thiazolyl)-2-methoxyiminoacetyl]amino]-2-carboxy-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-en-3-yl]-4-methyl-2-pyridinemethanol.
Purification: The ceftazidime intermediate is purified by various methods such as crystallization and chromatography.
Synthesis of Ceftazidime:
The ceftazidime intermediate is coupled with D-arginine and D-arginine amide using carbodiimide reagents to form N-[(6R,7R)-7-[[(2Z)-2-(2-amino-4-thiazolyl)-2-methoxyiminoacetyl]amino]-2-carboxy-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-en-3-yl]-L-arginine and N-[(6R,7R)-7-[[(2Z)-2-(2-amino-4-thiazolyl)-2-methoxyiminoacetyl]amino]-2-carboxy-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-en-3-yl]-L-arginine amide, respectively.
The coupled products are then reacted with sodium hydroxide and sodium carbonate to form the final product, ceftazidime.
The final product is purified by various methods such as crystallization, chromatography, and recrystallization.
Ceftriaxone and other third-generation cephalosporin antibiotics are used to treat organisms that tend to be resistant to many other antibiotics. Due to emergent resistance, ceftriaxone should not be used for the treatment of Enterobacter infections. Before using ceftriaxone, it is important to determine the susceptibility of the bacteria. If sepsis is being considered, empiric therapy may be initiated prior to susceptibility testing.
Medical uses include:Lower respiratory tract infections.Acute bacterial otitis media.Skin and skin structure infections.Urinary tract infections.Uncomplicated gonorrhea.Pelvic inflammatory.Disease.Bacterial sepsis.Intra-abdominal infections.Meningitis.Surgical prophylaxis.Lyme disease.
Ceftriaxone is also a choice drug for treatment of bacterial meningitis caused by pneumococci, meningococci, Haemophilus influenzae, and "susceptible enteric Gram-negative rods, but not Listeria monocytogenes."
In combination with doxycycline or azithromycin, ceftriaxone used to be recommended by the United States Centers for Disease Control and Prevention (CDC) for the treatment of uncomplicated gonorrhea. Due to increased risk of developing azithromycin resistant strains and the high efficacy of higher doses of ceftriaxone the guidance has been updated to mono-antibiotic therapy with a higher dose of ceftriaxone.
Pharmacokinetics Ceftriaxone
Absorption: Ceftriaxone can be administered intravenously and intramuscularly, and the drug is completely absorbed. It is not available orally.
Distribution: Ceftriaxone penetrates tissues and body fluids well, including cerebrospinal fluid to treat central nervous system infections.Ceftriaxone is reversibly bound to human plasma proteins and the binding of ceftriaxone decreases with increasing concentration from a value of 95% at plasma concentrations less than 25 mcg/mL to 85% at plasma concentration of 300 mcg/mL. Over a 0.15 to 3 g dose range in healthy adult subjects, the apparent volume of distribution ranged from 5.8 to 13.5 L.
Metabolism: 33–67% of ceftriaxone is renally excreted as unchanged drug, but no dose adjustments are required in renal impairment with dosages up to 2 grams per day. The rest is excreted in the bile as unchanged drug[30] which is ultimately excreted in feces as inactive compounds from hepatic and gut flora metabolism.
Elimination: The average elimination half-life in healthy adults is 5.8–8.7 (mean 6.5) hours, with some reviews estimated half-life up to 10 hours. In people with renal impairment, the average elimination half-life increases to 11.4–15.7 hours.
Our Factory
We have stable and superior route of synthesis, strict quality control and quality assurance system, experienced and responsible team, efficient and safe logistics. Based on this, our products are well recognized by the customers in Europe, Americas, Asia, Middle East etc.



FAQ
As one of the leading pharmaceutical intermediates manufacturers and suppliers in China, we warmly welcome you to buy high quality pharmaceutical intermediates made in China here from our factory. All chemicals are with high quality and competitive price.
Pharmaceutical Intermediates, EHATA, Thiotriazinone-
MXC-BDMAEEName: BIS(2-DIMETHYLAMINOETHYL) ETHER(A-1)read more
Cas no.: 3033-62-3
Purity: ≥99%
Appearance: Clear,...









